

The clear and permanently legible identification of medical products is no longer a competitive advantage today — it is a regulatory necessity.
Our new ultrashort-pulse laser marking system was developed precisely to meet these requirements and sets new standards in irreversible laser marking for medical instruments, surgical equipment, implants, and complex medical components.
Perfect marking without material impact
By using state-of-the-art ultrashort-pulse laser technology (pico-/femtosecond range), marking is performed with virtually no thermal influence (“cold ablation”). Even sensitive materials such as stainless steel, titanium, cobalt-chromium alloys, ceramics, or coated surfaces are permanently marked without microstructural changes, microcracks, or impairment of biocompatibility.
The result:
high-contrast, microscopically precise markings
excellent readability for camera- and scanner-based systems
absolute resistance to sterilization processes, chemicals, and mechanical stress


The system meets all requirements for UDI marking in accordance with MDR (EU) 2017/745 and FDA regulations.
The integrated system software provides direct access to global UDI databases (GUDID / EUDAMED-compatible data structures), enabling end-to-end, audit-proof traceability across the entire product lifecycle.
Key features at a glance:
automatic generation and management of UDI-DI and UDI-PI
direct database connectivity without media discontinuities
validation and audit support for quality and regulatory processes
seamless integration into existing MES and ERP systems
Designed for validated production environments
The laser marking system is consistently engineered for use in GMP-adjacent, validated manufacturing environments. Reproducibility, process stability, and comprehensive documentation are at the core of the system design.
Typical applications:
surgical instruments and instrument sets
orthopedic and dental implants
minimally invasive tools
medical precision components with micro-marking


With our ultrashort-pulse laser marking system “made in Germany,” you are not only investing in highly precise marking technology, but also in regulatory certainty, process stability, and long-term future readiness.
Permanent. Traceable. Regulatory-safe.
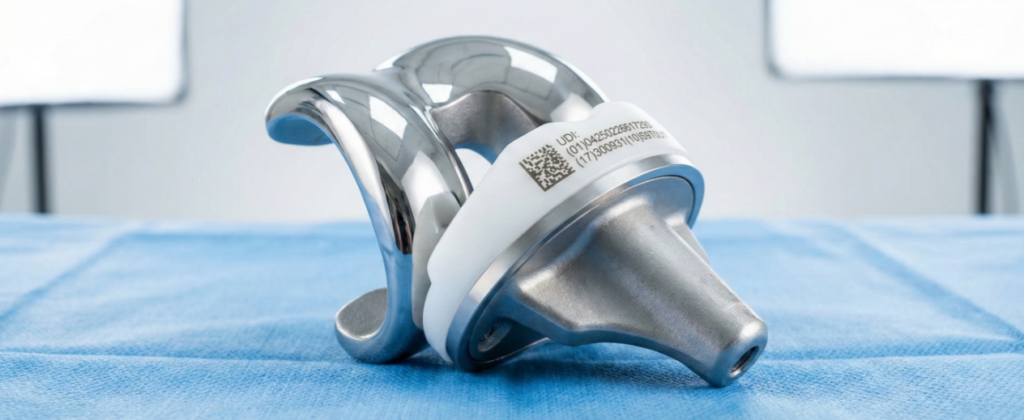

Unique Device Identification (UDI) is a core element of international medical device regulation and is mandatory for almost all medical devices under EU MDR (EU) 2017/745 as well as U.S. FDA requirements.
From the very beginning, our ultrashort-pulse laser marking system has been designed to fully meet all relevant UDI requirements and to ensure long-term regulatory compliance.
Permanent direct marking (Direct Part Marking, DPM)
For reusable medical instruments and implants, regulators require permanent, lifetime identification directly on the product. Ultrashort-pulse laser technology enables highly precise Direct Part Marking (DPM) with DataMatrix codes, human-readable text, and individual serial numbers—remaining permanently legible throughout the entire product lifecycle.
The markings meet all requirements for:
resistance to cleaning and sterilization cycles (e.g., steam, plasma, chemical processes)
mechanical and chemical durability
machine readability in accordance with ISO/IEC 16022 (DataMatrix)
high contrast grades for reliable verification in compliance with ISO/IEC 15415


The system software supports the complete UDI workflow – from data generation through international registration.
Via direct connectivity to global UDI databases, product and production data can be managed in a structured manner and documented in an audit-proof way.
Supported functions include:
management of UDI-DI (Device Identifier) and UDI-PI (Production Identifier)
automatic serial number, batch, and date management
structured data records aligned with GUDID (FDA) and EUDAMED logic
export and interface functions for MES, ERP, and QMS systems
Validation capability and audit security
For medical device manufacturers, the ability to validate the marking process is essential. The system meets the requirements for IQ/OQ/PQ concepts, reproducible marking results, and complete process documentation.
This enables support for:
regulatory audits in accordance with MDR and FDA
traceability in compliance with ISO 13485
risk-based process controls as defined in ISO 14971
Future-proof in an evolving regulatory landscape
Thanks to its modular software architecture, the laser marking system is fully update-capable and can be adapted to future regulatory changes in the UDI environment—an essential advantage given the ongoing expansion of EUDAMED and international database requirements.
The compact HAWK 2 laser processing station offers an exceptional level of flexibility and performance.
The use of state-of-the-art laser technology with outstanding beam quality ensures stable operation and the highest quality standards—even under full production conditions.
Depending on the application, the laser system can be equipped with a fiber laser, a diode-pumped solid-state laser, or a CO₂ laser.